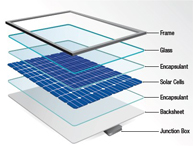
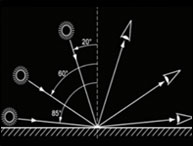
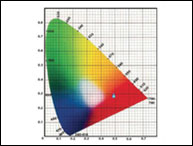
Instrument validation in the pharmaceutical industry
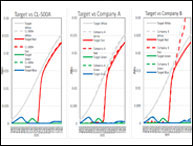
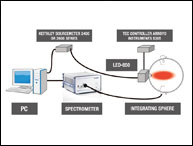
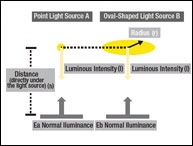
The pharmaceutical industry is one of the strictest industries to perform GMP (good manufacturing practice). For pharmaceutical companies to maintain a high GMP quality, proper qualification and validation protocols must be in place for the highly regulated pharmaceutical industry. Validation for an instrument is essential for maintaining the high-quality production of pharmaceutical products, and ensuring proper color measurement is necessary. Validation of an instrument usually consists of Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ).